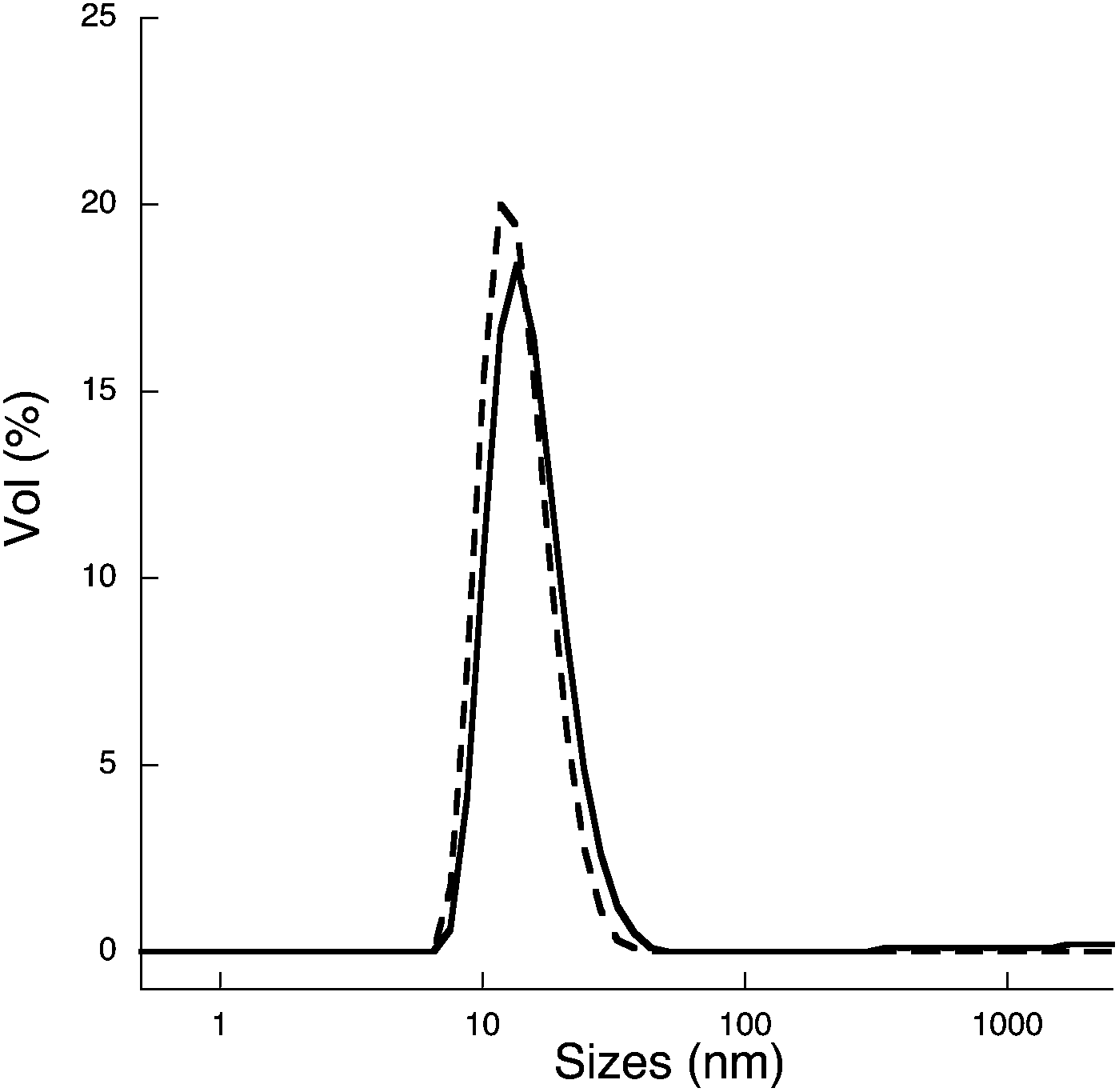
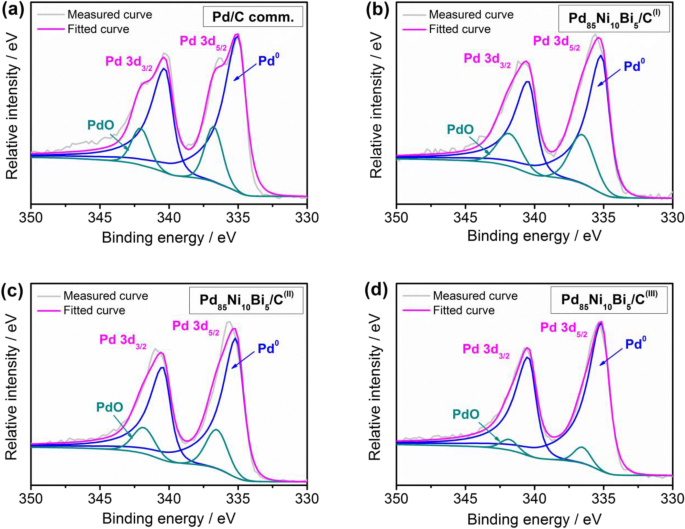
5,6 Rh has high catalyst activity for example, NOx reduction catalysts, 7,8 but it is one of the most expensive elements. No combination of binary alloy from these elements (Ru–Rh, Rh–Pd and Ru–Pd) forms a solid-solution alloy throughout the whole composition range at room temperature. 1–4 Pd, Rh and Ru are neighboring noble elements in the 4d transition metal series. Introduction Pt, Pd, Rh and Ru have been used as three-way catalysts, and Pd is well-known as a hydrogen storage metal. The solid-solution structures and local structure were confirmed via HR-TEM, X-ray photoelectron spectroscopy (XPS) and X-ray absorption fine structure spectroscopy (XAFS). The lattice parameters of nanoparticles followed the Vegard's law, and the energy-dispersive X-ray spectroscopy (EDX) results almost coincided with those obtained for the starting bulk mixtures. The particle sizes of the floated and sedimented samples were measured to be <10 and <20 nm, respectively, via high-resolution transmission electron microscopy (HR-TEM). We have synthesized solid-solution nanoparticles (Pd : Ru = 1 : 3, 1 : 1 and 3 : 1) in an immiscible Pd–Ru system by the pulsed plasma in liquid method using Pd–Ru mixture bulk electrodes.
